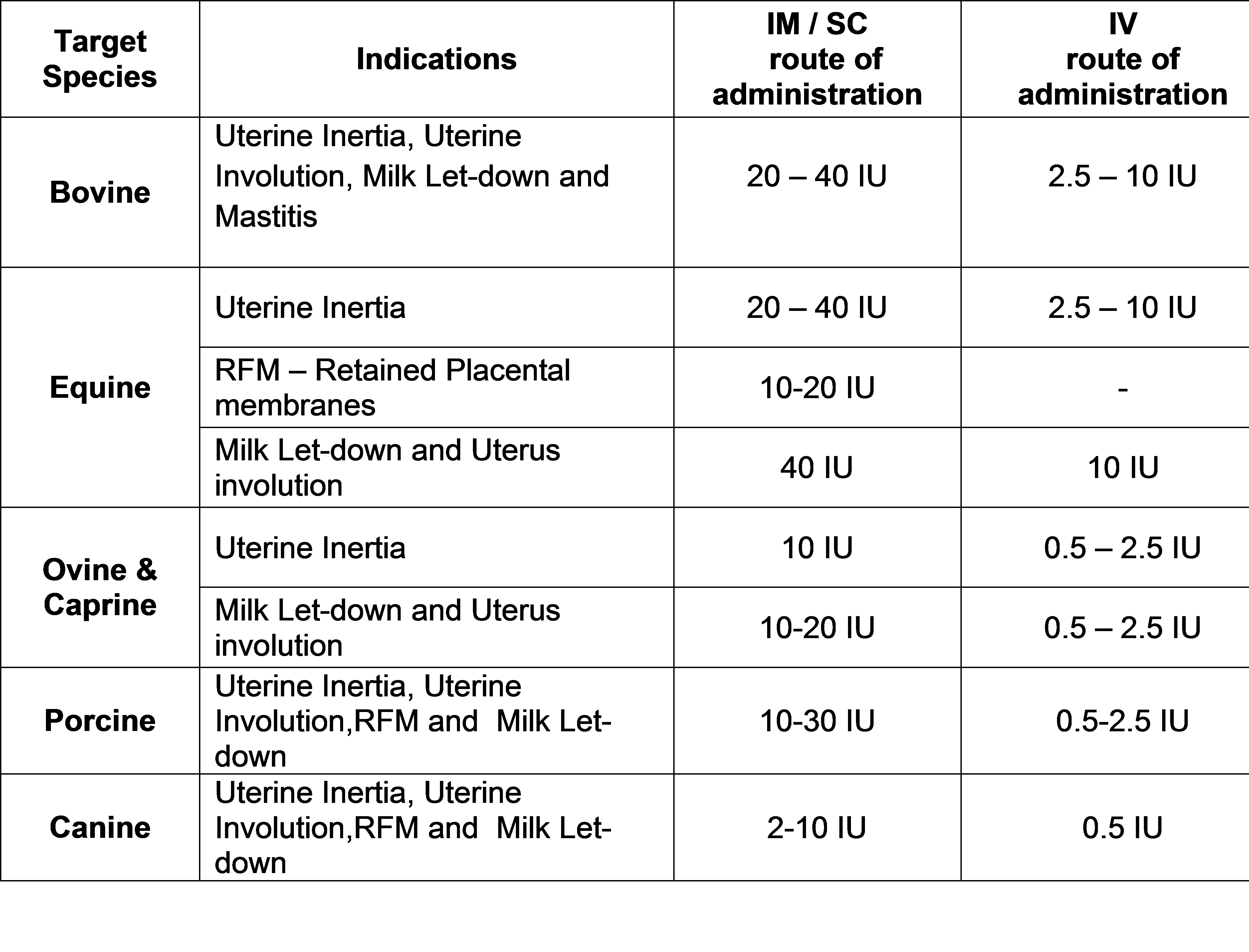KARNTAKA ANTIBIOTICS AND PHARMACEUTICALS LIMITED
(Government of India Enterprise)
Karnataka Antibiotics & Pharmaceuticals Limited(KAPL) (A Govt. of India Enterprise), Bengaluru, Karnataka- 560058 is engaged in manufacturing and marketing of various Pharmaceutical /Ayurvedic / Veterinary and Agro-chemical products.
To cater the requirement of customers and the company, KAPL intends to appoint Clearing and Forwarding Agents ( C & F Agent) in the state of TELENGANA (for Telangana & Andhra Pradesh)
.
Notice Inviting Tender (NIT)
For Appointment of C & F Agency for Telangana & Andhra Pradesh
Tender No: KAPL/MKT/2025/ C & F AGENT/03 DT. 15.09.25
| Last date & time for submission of Tender | (21 days from the date of Publication in KAPL website) up to 18:00 Hrs |
| Date and time of opening of Bids | 07.10.25 at 10.30 Hrs |
- For Details Click here
- For Annexures A, B & C Click Here
- For General Instructions Click Here
- For Integrity Agreement Click Here
*******************************************************************************************************************************************************************