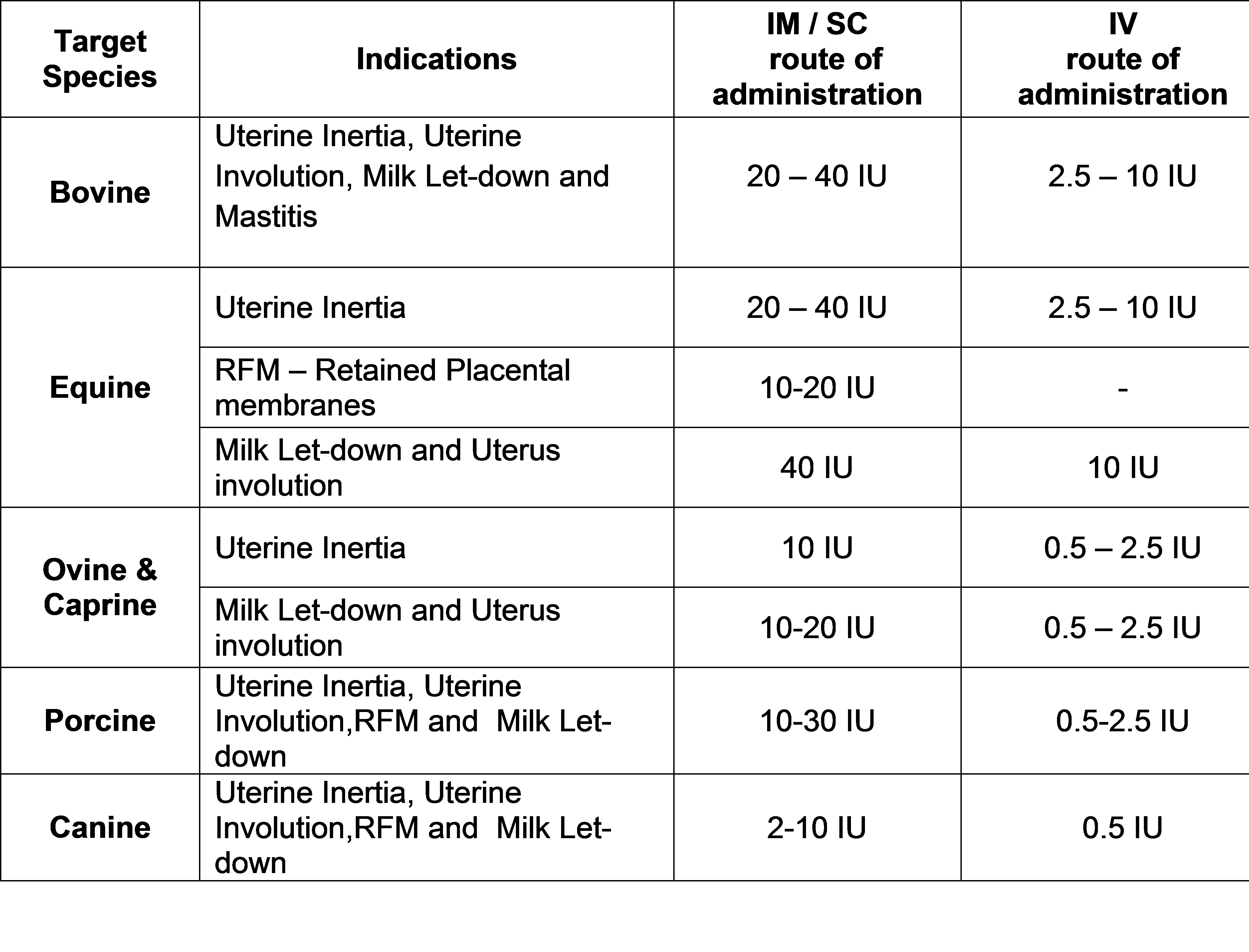R&D Policy:
To Develop Drug Products with an emphasis on Integrity, Quality, Process Robustness and Stability at an affordable cost to the patients world-wide, in alignment with organizational goals and objectives.
TEAM :
KAPL Research and Development Team is championed and mentored by Managing Director.
Under the leadership and vision, the Formulation Development has evolved into a Research and Development Unit, to align with organization’s mission.
Team R&D comprises of Formulation, Analytical and Packaging Development Scientists with expertise to develop, optimize and analyze various drug delivery systems.